
State-of-the-art nanotechnology improving life for diabetes patients
Technology
Lifecare’s miniaturized sensor technology measures changes by reading osmotic pressure variations in a small chamber. While Lifecare’s initial focus is on glucose monitoring, the sensor technology is adaptable for use in a wide range of medical applications, including diagnostics and biomarker monitoring. This broad applicability provides Lifecare with future opportunities for expansion into new areas of healthcare.

While current technology for continuous glucose monitoring uses glucose oxidase or flouresence as sensing principles, Lifecare’s technology is based on the osmotic pressure. We believe that our technology offers greater stability and longevity compared to glucose oxidase and fluorescence-based glucose monitoring, as it does not rely on enzymes or chemical dyes that degrade over time. It is less susceptible to biofouling and external interferences, improving accuracy in continuous monitoring. Additionally, our osmotic pressure sensor will be more energy-efficient, making it well-suited for long-term implantable glucose monitoring solutions
The osmotic pressure technology
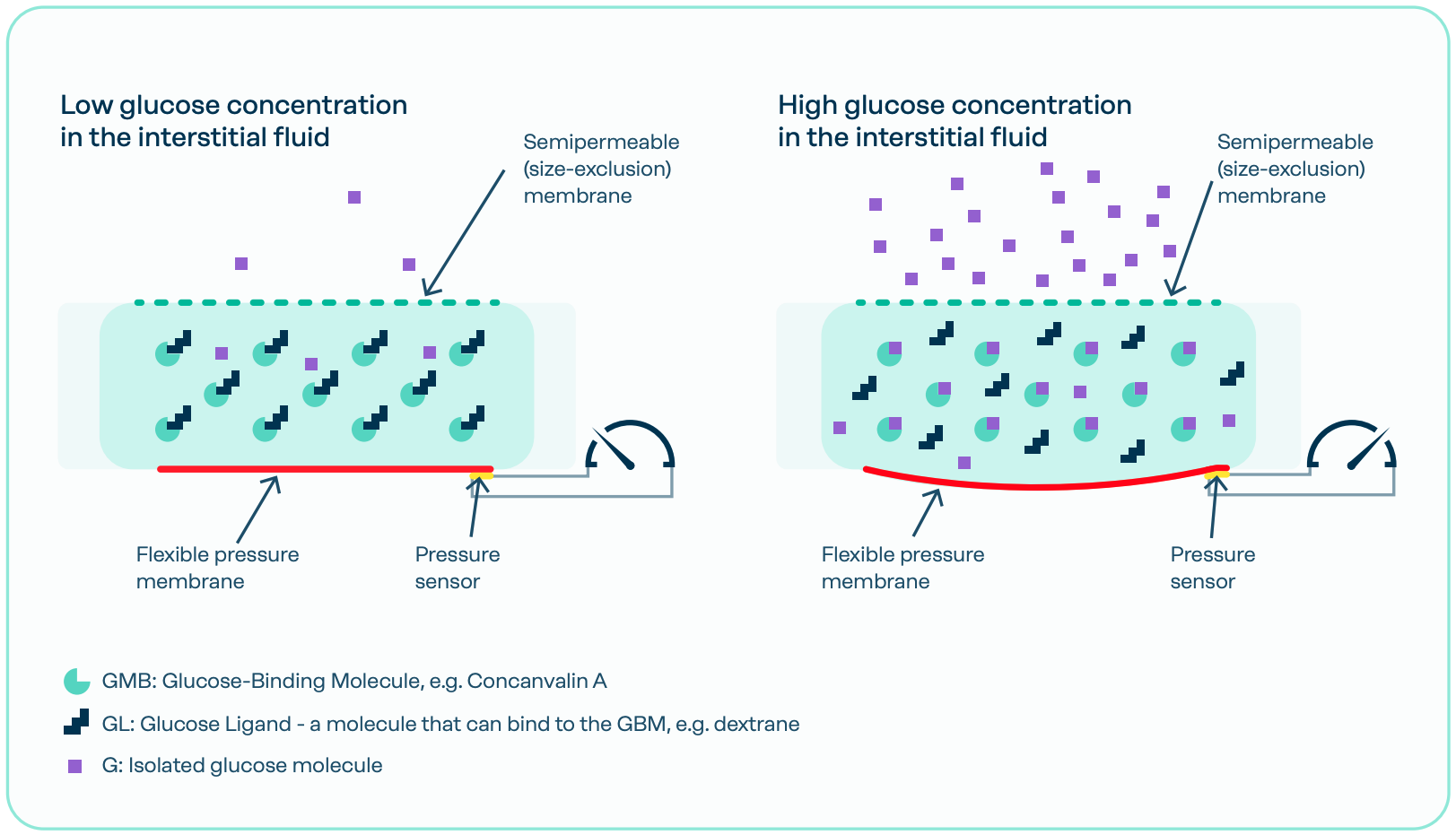
Lifecare’s osmotic pressure technology is based on biochemical reactions where glucose connects to molecules in a closed chamber. This reaction generates a pressure variation that can be measured and monitored for glucose sensing purposes.
The process is fully reversible. As glucose concentrations decrease, glucose molecules detach from the binding sites, causing the osmotic pressure to decline. There is a linear relationship between the glucose concentration in the surrounding fluid and the measurable osmotic pressure in the chamber. Additionally, since the technology does not consume any molecules when generating the signal, it has the potential for long-term use within the body.
Lifecare holds several important patents that are central to its innovative glucose monitoring technology. Read more about the patents here and in Note 11 in the Financial Statements.
When glucose penetrates the semipermeable membrane into the chamber, glucose ligands (GL) are released from the glucose binding molecule (GBM) binding sites. This occurs because glucose has a slightly higher binding affinity to the GBM receptor. As a result, each glucose molecule causes the release of a GL molecule.

The newest branches of science
Nanosciences and -technology belong to the newest branches of science and are regarded by many researchers as key technolog…
Nanosciences and -technology belong to the newest branches of science and are regarded by many researchers as key technologies of this early 21st century. By manipulation of materials on a scale of down to one nanometer (one millionth of a millimeter), the historically split sciences of physics, chemistry, biology and medicine become all intertwined, making nanoscale technologies a unique field of interdisciplinary research. The extreme sensitivity of a cantilever in terms of mechanic deflection can be used for its application as a sensor.
Based on patented method for 3D printing using focused electron beam technologies, Lifecare orchestrates physical, biological and chemical properties of device components, fluids and mechanics in the nanoscale range to achieve sensing properties with high resolution.
3D Nanoprinting enables printing of sensors on any transducer substrate regardless of their thickness or material. In fact, the method allows printing of sensors in sizes down to few nanometers (one millionth of a millimeter) without reducing the sensitivity.


